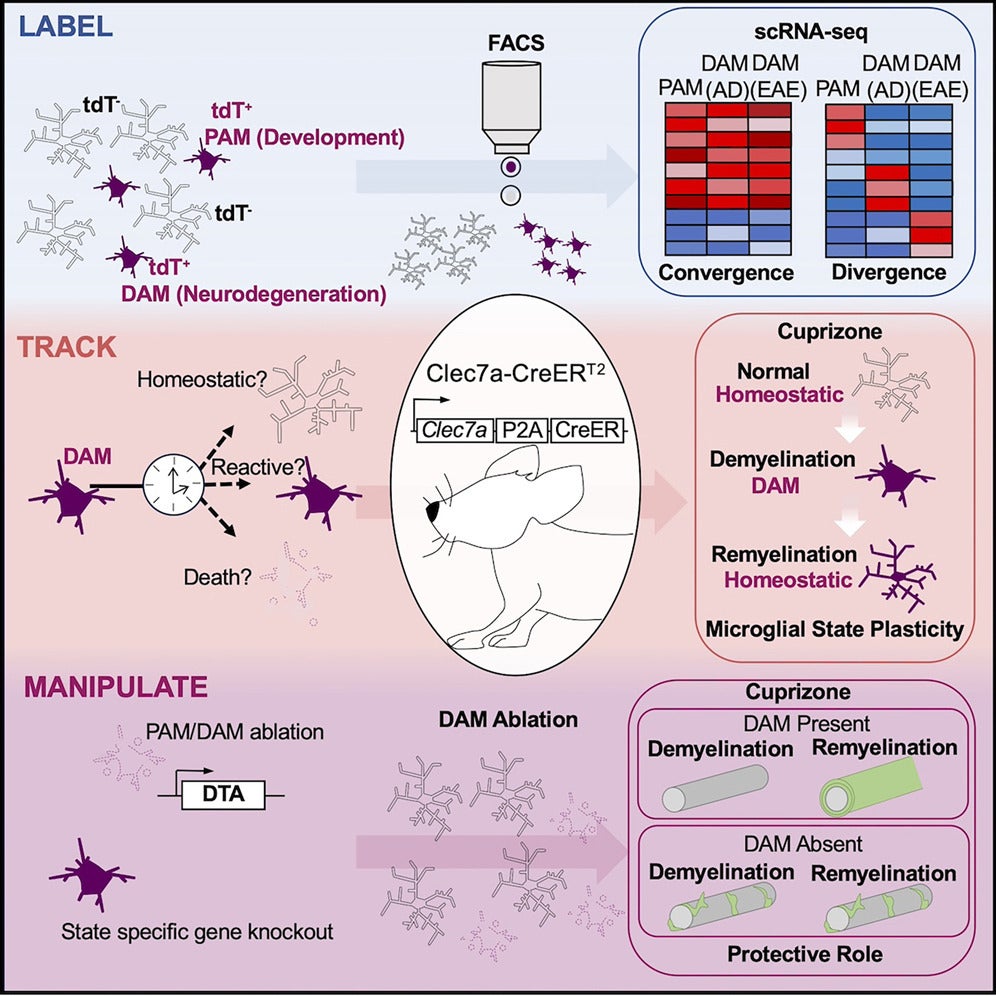
A recent study published in Immunity by the Li lab opens a new chapter for the study of microglial function in development and neurodegeneration with remarkable precision. Microglia are now recognized as critical functional modulators of brain development and neurodegeneration. Recent single-cell transcriptomic studies have identified important microglial states such as proliferative region-associated microglia (PAM) and disease-associated microglia (DAM) defined by the expression of unique gene signatures. However, for a long time, there were no tools to study microglia cells in a state-specific manner. The Li lab developed a novel inducible genetic tool, Clec7a-CreERT2, which allows scientists, for the first time, to efficiently and specifically label these microglial states both acutely and over the long term. This provides numerous opportunities to understand the role of microglia in how they shape the nervous system and how they respond to disease. The paper was also highlighted in a commentary review in Immunity.
This transgenic mouse line, Clec7a-CreERT2, is highly versatile. It is designed based on the expression of the shared marker gene, Clec7a, in microglia from various developmental and disease contexts, which allows specific targeting of these microglial cell populations. With this mouse line scientists can visualize the microglia cells in different contexts as well as track and functionally manipulate them. Being able to tag the cells at one point in time and assess them at a later point is particularly attractive for scientists, because it reveals to what extent microglia are plastic and how they dynamically change in disease settings where both protective and maladaptive functions of microglia are implicated.
This paper highlights the application of this tool to label PAM during early postnatal development, and DAM in a mouse model of Alzheimer’s disease and two models of multiple sclerosis. Systematic comparisons of these microglial states that share similar signatures enabled the authors to define converging and context-dependent genes and pathways.
“Perhaps, the most exciting discovery of our study is that we provide in vivo evidence demonstrating the remarkable plasticity of reactive microglial states,” says Dr. “Tristan” Qingyun Li, the corresponding author of this paper.
In addition to tracking microglia over time, scientists will also be able to specially remove DAM in a disease model or create specific gene knockout to gain functional insights. This will lay a foundation for future development of precisely targeted, microglia-based therapies.
“We believe this model can be used in a variety of disease contexts that we didn’t particularly study in the paper.” says Kia Barclay, a PhD student in the Li lab and the first author of the paper, “Taking the DAM state as an example, it appears to be a conserved response in aging and many other neurodegenerative diseases, such as Parkinson’s disease and Amyotrophic lateral sclerosis.”
Since the publication of this paper, scientists around the world have reached out to the lab and requested the mouse line for collaborations. “We have shown the neuroprotective function of DAM during remyelination (in the multiple sclerosis model). This is just scratching the surface.” says Dr. Li. Having removed a major bottleneck, the lab has geared up to tackle some long-standing mysteries about microglia in brain development and disease.
Featured in this article:
Kia Barclay

Kia is a fourth-year graduate student in the neuroscience program at Washington University in St. Louis. She earned her Bachelor of Arts in Neuroscience from Wellesley College in 2019. Alongside her research focus on microglia biology in white matter disease, Kia is deeply committed to serving underrepresented communities through science outreach and education. She hopes to encourage others to embrace the values of resilience, curiosity, and the individuality inherent in one’s scientific journey.
Li Lab

The Li laboratory is broadly interested in neuroimmunology with a focus on microglial biology. Particularly, the lab is interested in combining cutting-edge single-cell genomic technologies with in vitro and in vivo genetic, molecular and cellular tools to investigate microglial development, heterogeneity and mechanisms of neuro-immune interactions underlying brain structure and disease. We try to address two major questions: (1) how microglia (and other immune cells) are different in development, homeostasis and aging, and related to that, how these different populations of microglia interact with other neural cells to control brain development and functioning; (2) how microglial fate is specified and diverged from other tissue macrophages during early embryonic development. By studying these questions, the overarching goal is to gain a better understanding of microglial functions in the establishment of the nervous system, as well as how changes in these functions contribute to neurological diseases.